RFP LAB

Purpose:
- Make RFP from jellyfish bacteria
- Learn about steps genetic engineering
Materials + Procedure:
- 2a- materials + procedure can be found in Amgen lab manual part 2a
- 4a- materials + procedure can be found in Amgen lab manual part 4a
- 5a- materials + procedure can be found in Amgen lab manual part 5a
- 6a- materials + procedure can be found in Amgen lab manual part 6a
Experimental Overview:
Part 2a: We first needed to verify the plasmid by using restriction digest. Then we cut the plasmid using BamHI and Hind III to cut out the RFP-ara.
Part 4a: In this part we verified the plasmid digest by electrophoresis.
Part 5a: Next we transformed the bacteria with the recombinant plasmid.
Part 6: Finally we purified the RFP using chromatography.
Part 2a: We first needed to verify the plasmid by using restriction digest. Then we cut the plasmid using BamHI and Hind III to cut out the RFP-ara.
Part 4a: In this part we verified the plasmid digest by electrophoresis.
Part 5a: Next we transformed the bacteria with the recombinant plasmid.
Part 6: Finally we purified the RFP using chromatography.
Copy Paste |
chrome-extension://bpmcpldpdmajfigpchkicefoigmkfalc/views/app.html
|
Results
All lab Questions can be found in the lab manual links above. All the answers to the questions can be found below.
Data Results
Before the 2a Lab:
1. If pARA-R is digested with BamHI and HindIII, what fragments are produced? Record the nucleotide sequence of the sticky ends and the length of each fragment (bp), and indicate the genes and other important sequences present on each fragment.
Two fragments are produced. They are RFP with pBAD and Ara-C with ori with Amp-R. The RFP plus pBAD is 807 BP, and Ara-C, ori, and Amp-R is 4495 BP.
2. In order to create a plasmid that can produce the red fluorescent protein in
bacteria, what components are needed in the plasmid?
We need the RFP gene and Ara-C.
3. If the uptake of DNA by bacteria is inefficient why is a selectable marker critical in cloning a gene in bacteria? The selectable marker allows only the desired bacteria to grow.
Separates the bacteria from the desired gene.
2a Questions:
1. List in words or indicate in a drawing the important features of a plasmid vector that are required to clone a gene. Explain the purpose of each feature. Ori = origin of replication; RFP = red fluorescent protein (gene of interest); Amp-R = selectable marker; Ara-C = binds to promoter so we get transcription of gene of interest.
2. What role do restriction enzymes have in nature? Restriction enzymes are a defense mechanism.
Cut up foreign bodies.
3. Using your understanding of evolution, why would bacteria retain a gene that gives them resistance to antibiotics? How is the existence of bacteria with antibiotic resistance affecting medicine today?
Bacteria retain genes that give them resistance to antibiotics to protect themselves from disease.
4. Bacteria, sea anemones, and humans seem, on the surface, to be very different organisms. Explain how a gene from humans or a sea anemone can be expressed in bacteria to make a product never before made in bacteria.
Central dogma is the same in all organisms.
5. Due to a mishap in the lab, bacteria carrying a plasmid with an ampicillin resistant gene and bacteria carrying a plasmid with a gene that provides resistance to another antibiotic (kanamycin) were accidentally mixed together. Design an experiment that will allow you to sort out the two kinds of bacteria. Create a petry dish that grows both Kan and Amp bacteria.
Put half of the mixed bacteria into each petry dish and wait a day. The bacteria should have died and then you should be able to separate them apart.
4a Questions:
1. The purpose of growing bacteria that resists ampicillin is to make sure that the desired cell lives.
2. If the bacterial cells are not given arabinose, then the pARA-R plasmid will not turn on the promoter. Then the protein will not turn red.
3. In the LB plate, both the P- and P+ will grow. In the LB/Amp plate, no P- will grow, but P+ will. In the LB/ Amp/Ara plate, there will barely any bacteria growth.
5a Questions:
1. Our predictions sort of matched our results. The LB plate had limited growth. The LB/Amp and LB/Amp/Ara plate matched our prediction.
2. There were no red colonies visible either due to temperature or not enough time in the incubator.
3. The LB-amp-ara plate prevents transcription to occur, which allows RFP to be expressed. The LB-amp does not have Ara.
4. It is important to have multiple copies so there is a greater chance of the promoter being turned on.
5. The RFP gene is expressed as a trait through transcription.
6a Questions:
1. The red fluorescent protein can be seen separated because of its red cells.
2. The supernatant is clear liquid the pellets are pink.
6b Questions:
1. Binding Buffer (BB): causes amino acid and protein bind to the resin beads.
i
Wash Buffer (WB): removes loose proteins that are not bound to the resin beads.
Elution Buffer (EB): takes off protein off resin beads.
Column Equilibration Buffer (CEB): stores resin beads.
2. This time, the supernatant was more pink than clear. The pellet was a little darker pink than the supernatant.
1. If pARA-R is digested with BamHI and HindIII, what fragments are produced? Record the nucleotide sequence of the sticky ends and the length of each fragment (bp), and indicate the genes and other important sequences present on each fragment.
Two fragments are produced. They are RFP with pBAD and Ara-C with ori with Amp-R. The RFP plus pBAD is 807 BP, and Ara-C, ori, and Amp-R is 4495 BP.
2. In order to create a plasmid that can produce the red fluorescent protein in
bacteria, what components are needed in the plasmid?
We need the RFP gene and Ara-C.
3. If the uptake of DNA by bacteria is inefficient why is a selectable marker critical in cloning a gene in bacteria? The selectable marker allows only the desired bacteria to grow.
Separates the bacteria from the desired gene.
2a Questions:
1. List in words or indicate in a drawing the important features of a plasmid vector that are required to clone a gene. Explain the purpose of each feature. Ori = origin of replication; RFP = red fluorescent protein (gene of interest); Amp-R = selectable marker; Ara-C = binds to promoter so we get transcription of gene of interest.
2. What role do restriction enzymes have in nature? Restriction enzymes are a defense mechanism.
Cut up foreign bodies.
3. Using your understanding of evolution, why would bacteria retain a gene that gives them resistance to antibiotics? How is the existence of bacteria with antibiotic resistance affecting medicine today?
Bacteria retain genes that give them resistance to antibiotics to protect themselves from disease.
4. Bacteria, sea anemones, and humans seem, on the surface, to be very different organisms. Explain how a gene from humans or a sea anemone can be expressed in bacteria to make a product never before made in bacteria.
Central dogma is the same in all organisms.
5. Due to a mishap in the lab, bacteria carrying a plasmid with an ampicillin resistant gene and bacteria carrying a plasmid with a gene that provides resistance to another antibiotic (kanamycin) were accidentally mixed together. Design an experiment that will allow you to sort out the two kinds of bacteria. Create a petry dish that grows both Kan and Amp bacteria.
Put half of the mixed bacteria into each petry dish and wait a day. The bacteria should have died and then you should be able to separate them apart.
4a Questions:
1. The purpose of growing bacteria that resists ampicillin is to make sure that the desired cell lives.
2. If the bacterial cells are not given arabinose, then the pARA-R plasmid will not turn on the promoter. Then the protein will not turn red.
3. In the LB plate, both the P- and P+ will grow. In the LB/Amp plate, no P- will grow, but P+ will. In the LB/ Amp/Ara plate, there will barely any bacteria growth.
5a Questions:
1. Our predictions sort of matched our results. The LB plate had limited growth. The LB/Amp and LB/Amp/Ara plate matched our prediction.
2. There were no red colonies visible either due to temperature or not enough time in the incubator.
3. The LB-amp-ara plate prevents transcription to occur, which allows RFP to be expressed. The LB-amp does not have Ara.
4. It is important to have multiple copies so there is a greater chance of the promoter being turned on.
5. The RFP gene is expressed as a trait through transcription.
6a Questions:
1. The red fluorescent protein can be seen separated because of its red cells.
2. The supernatant is clear liquid the pellets are pink.
6b Questions:
1. Binding Buffer (BB): causes amino acid and protein bind to the resin beads.
i
Wash Buffer (WB): removes loose proteins that are not bound to the resin beads.
Elution Buffer (EB): takes off protein off resin beads.
Column Equilibration Buffer (CEB): stores resin beads.
2. This time, the supernatant was more pink than clear. The pellet was a little darker pink than the supernatant.
Reflection
Things I liked about this project-
- Growing up the Bacteria from the petrey dish
- Learning how to extract and put in different proteins
- Collaborating with my group
- Not breaking our seal
- Making sure we have more of the protein
- Always on task
- Never had to stay in and finish
- Never argued
Lab 6:
Before the Lab:
1. How can solutions of different salt concentrations, which will unfold proteins to varying degrees, be used to help purify red fluorescent protein using column chromatography?
Proteins are unfolded in the highly salt concentrated buffer. The proteins that don't flow out the coulmn are refolded when lower salt concentrations are added, thus they leave the column.
Lab 6 Questions:
1. Why is a protein’s conformation important for carrying out its function?
A protein's conformation is important because it determines the promoter regions of the given protein. These promoter regions are what give each protein their different functions.
2. What properties of the amino acids in a protein relate to protein folding?
The sequence of an amino acid determines how it will fold into a protein.
3. Does the eluate containing your red fluorescent protein appear less bright or brighter than it did in the cell lysate following centrifugation? If there is a noticeable difference in the intensity of the red color, what might account for that?
The RFP-containg elute is brighter than the cell lysate. This is because the elute contains the most RFP--the cell lysate contained all the cell proteins.
4. What characteristic of red fluorescent protein is used as the basis for separation by column chromatography?
The red fluorescent protein sticks to the resin column when unfolded (more hydrophobic amino acids).
5. How might the column chromatography procedure be adjusted or modified to increase the purity of the red fluorescent protein sample?
If we repeated the process with more wash buffers and were more careful about collection, we could increase the red fluorescent protein purity.
ANALYSIS/CONCLUSION:In this lab, we attempted to make a glowing red bacteria colony. While they might not have been glowing, we did manage to grow a colony with the correct gene (red fluorescent protein). We did this by using restriction enzymes to isolate the RFP gene and then confirmed that our work was correct by using gel electrophoresis. Then we used recombinant plasmids and chromotography to seperate the red protein into a tube.
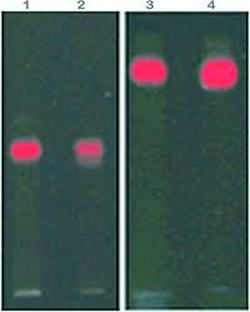
After extracting the RFP, we ran it in a gel:
Our particular column was the purple line on the first gel. We had very little protein in the proper places and our gel had some issues as seen in the odd formation. however when looking at 2nd and 3rd period's gels. We can see that some groups had really good RFP extraction. They had a nice thick band at the proper place according to the chart above.
<img src="http://sawyerikedastem.weebly.com/uploads/2/2/8/9/22896468/2020793_orig.jpg" alt="Picture"/>drlafevrebernt.weebly.com/uploads/1/1/8/1/11810063/amgen_sg_sequence2_rev2014.pdf
Before the Lab:
1. How can solutions of different salt concentrations, which will unfold proteins to varying degrees, be used to help purify red fluorescent protein using column chromatography?
Proteins are unfolded in the highly salt concentrated buffer. The proteins that don't flow out the coulmn are refolded when lower salt concentrations are added, thus they leave the column.
Lab 6 Questions:
1. Why is a protein’s conformation important for carrying out its function?
A protein's conformation is important because it determines the promoter regions of the given protein. These promoter regions are what give each protein their different functions.
2. What properties of the amino acids in a protein relate to protein folding?
The sequence of an amino acid determines how it will fold into a protein.
3. Does the eluate containing your red fluorescent protein appear less bright or brighter than it did in the cell lysate following centrifugation? If there is a noticeable difference in the intensity of the red color, what might account for that?
The RFP-containg elute is brighter than the cell lysate. This is because the elute contains the most RFP--the cell lysate contained all the cell proteins.
4. What characteristic of red fluorescent protein is used as the basis for separation by column chromatography?
The red fluorescent protein sticks to the resin column when unfolded (more hydrophobic amino acids).
5. How might the column chromatography procedure be adjusted or modified to increase the purity of the red fluorescent protein sample?
If we repeated the process with more wash buffers and were more careful about collection, we could increase the red fluorescent protein purity.
ANALYSIS/CONCLUSION:In this lab, we attempted to make a glowing red bacteria colony. While they might not have been glowing, we did manage to grow a colony with the correct gene (red fluorescent protein). We did this by using restriction enzymes to isolate the RFP gene and then confirmed that our work was correct by using gel electrophoresis. Then we used recombinant plasmids and chromotography to seperate the red protein into a tube.
After extracting the RFP, we ran it in a gel:
Our particular column was the purple line on the first gel. We had very little protein in the proper places and our gel had some issues as seen in the odd formation. however when looking at 2nd and 3rd period's gels. We can see that some groups had really good RFP extraction. They had a nice thick band at the proper place according to the chart above.
<img src="http://sawyerikedastem.weebly.com/uploads/2/2/8/9/22896468/2020793_orig.jpg" alt="Picture"/>drlafevrebernt.weebly.com/uploads/1/1/8/1/11810063/amgen_sg_sequence2_rev2014.pdf

